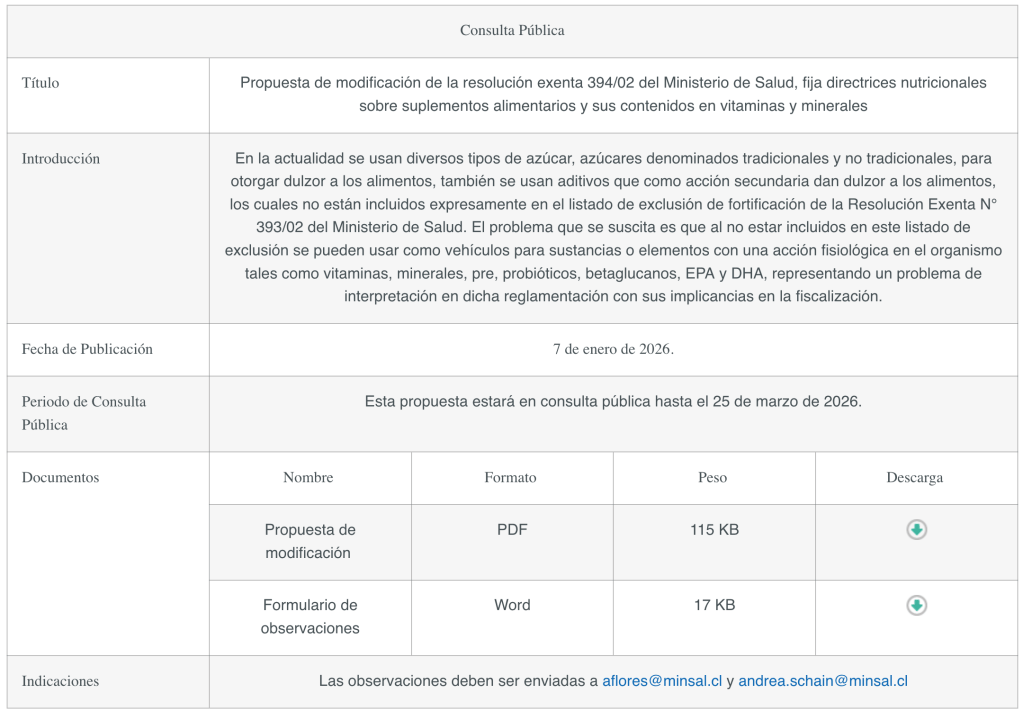
The Ministry of Health has published a proposed amendment to Exempt Resolution 394/02 of the Ministry of Health, which establishes nutritional guidelines for dietary supplements and their vitamin and mineral content.
Currently, various types of sugar—both traditional and non-traditional—are used to sweeten foods, as are additives that, as a secondary effect, impart sweetness, none of which are expressly included in the fortification exclusion list of Exempt Resolution No. 393/02 issued by the Ministry of Health. The problem that arises is that, since they are not included in this exclusion list, they can be used as vehicles for substances or elements with a physiological effect on the body, such as vitamins, minerals, prebiotics, probiotics, beta-glucans, EPA, and DHA, posing an interpretive challenge to the regulation and its enforcement.