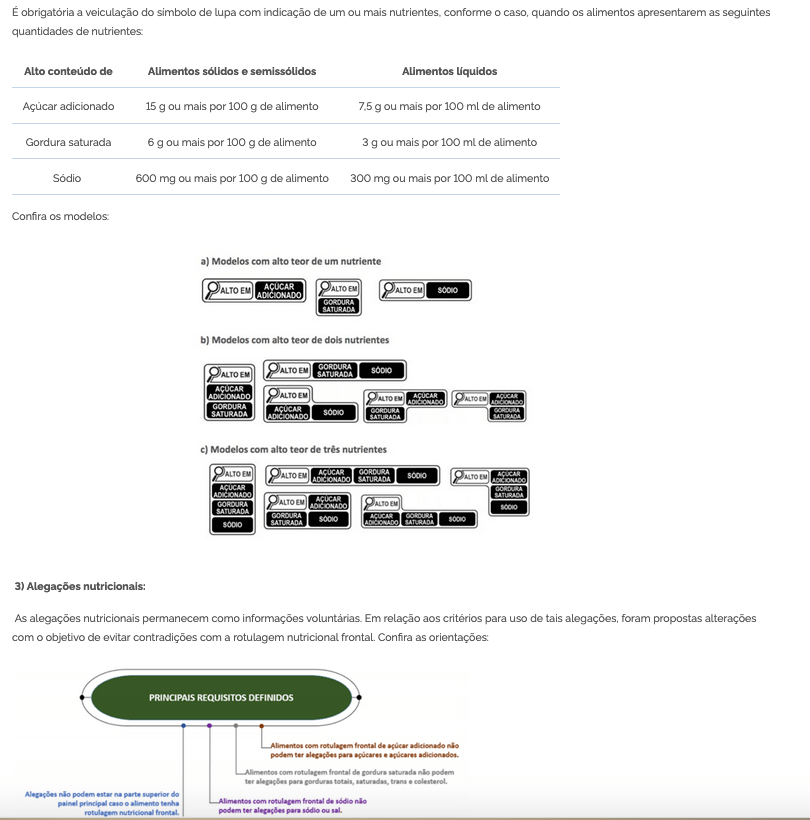
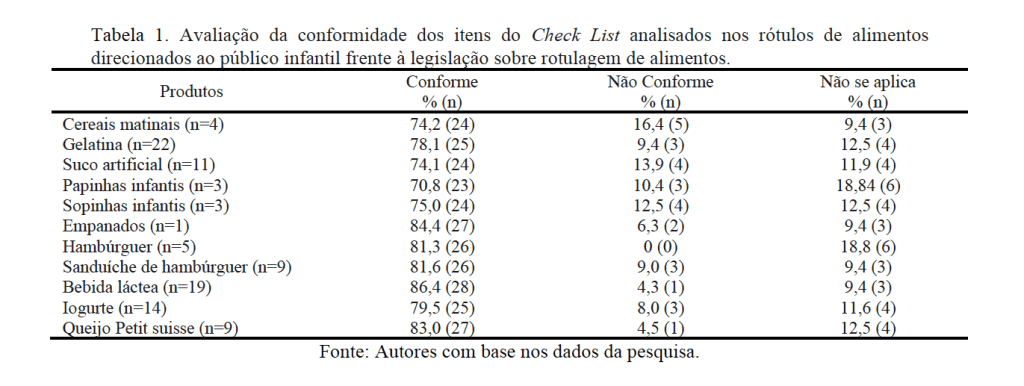
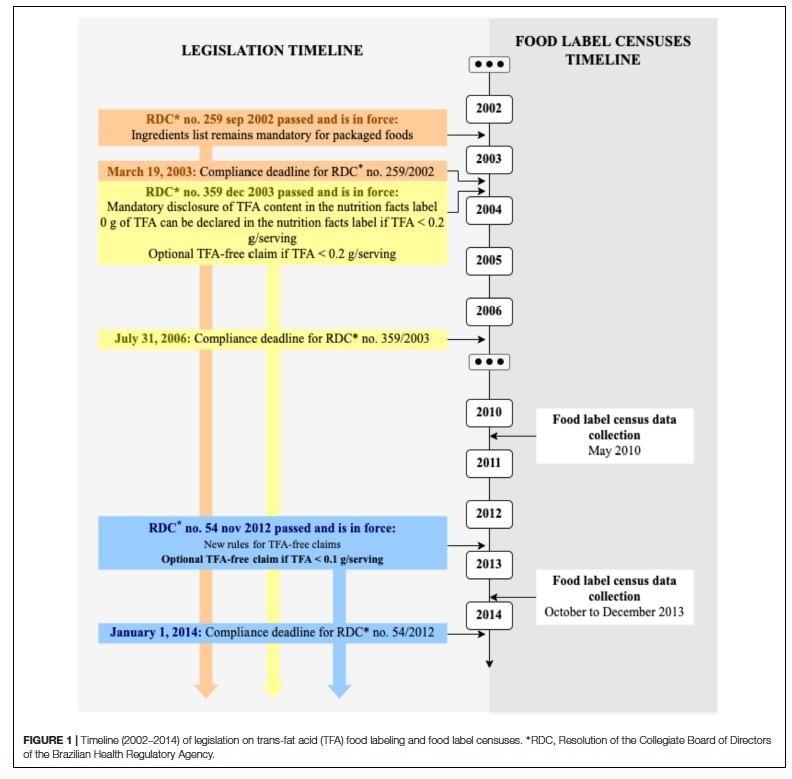
Bill 2257/22 was introduced in the Chamber of Deputies, which prohibits the use of titanium dioxide in the manufacture of food, as well as the importation of articles containing this substance. The text under analysis in the House establishes that any non-compliance with the regulations will be a violation of federal health regulations (Law 6.437/77).
Titanium dioxide is a chemical compound, derived from a natural mineral, used as an industrial colorant, among other purposes. Currently, this use is authorized in Brazil by the National Health Surveillance Agency (Anvisa).
According to the author of the proposal, Deputy Ney Leprevost (União-PR), in the case of the use of titanium dioxide in food, studies indicate the potential to cause inflammatory lesions in the intestinal tract of experimental animals.
A proposal with similar content (PL 1370/11) passed through the House for two legislatures and was even approved by the Committee on Environment and Sustainable Development, but ended up shelved in 2019.