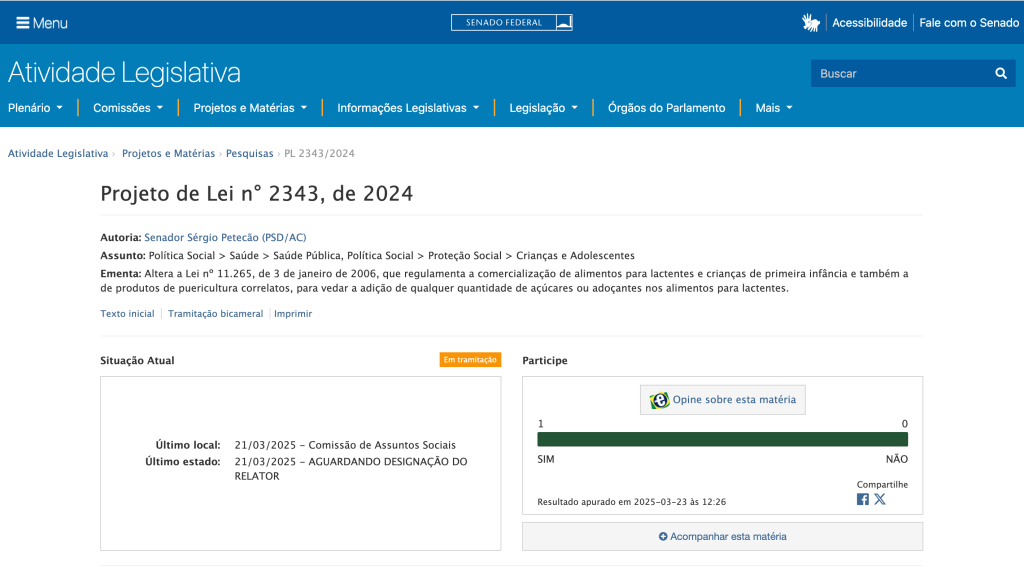
The Senate Committee on Human Rights and Participative Legislation (CDH in Portuguese) approved Bill 2.343/2024, which amends Law 11.265, which regulates the commercialization of food for infants and young children, prohibiting the addition of any amount of sugar and sweeteners to food intended for infants.
The bill by Senator Sérgio Petecão (PSD-AC) received the favorable vote of the rapporteur, Senator Laércio Oliveira (PP-SE), and now passes to the Social Affairs Committee (CAS) for analysis.