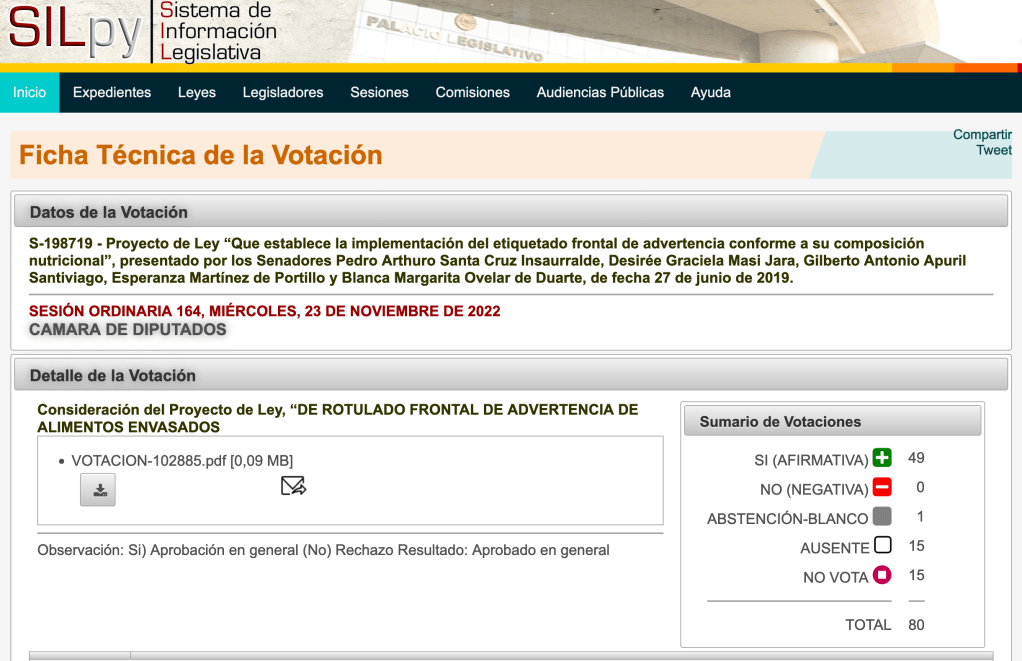
The Senate approved the law on front labeling of packaged foods, with the amendments introduced in the House of Representatives. With this version, products will no longer carry the inscription “high in calories”, since they will have the exact amount of calories.
The head of the Public Health Commission, Jorge Querey, insisted on the need to establish mandatory labeling, according to the nutritional composition of sugars, saturated fats and sodium, incorporating the information of the “high calorie” content, in a visible, quick and easily understandable way for the consumer’s decision making.