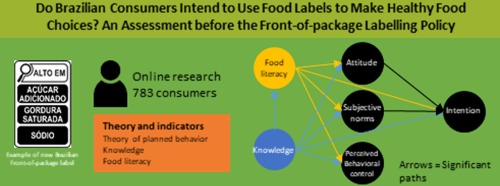
Front-of-package labelling is a measure that can facilitate consumption of healthier foods. This study examined the predictors of consumers’ intentions to use food labelling to make healthy food choices and prevent non-communicable diseases before the implementation of the front-of-package labelling policy in Brazil. An extended theory of planned behaviour incorporating knowledge and food literacy served as the theoretical foundation. First, experts (n = 6) and the target population (n = 30) validated the questionnaire, and the validation data were analysed using content validity coefficients (CVC). An online questionnaire was administered to 783 participants, and data were collected between May and October 2022. Partial least squares structural equation modelling (PLS-SEM) was used to test the prediction hypothesis. The questionnaire showed adequate content validity, with all indicators of CVC ≥ 0.75. In the PLS-SEM attitudes (p < 0.001), subjective norms (p < 0.001), knowledge (p < 0.001), and food literacy (p < 0.001) positively predicted the intention to use food labels to make healthy food choices, confirming the main hypothesis. Also, Food literacy and knowledge predicted all constructs of the theory of planned behaviour (p < 0.001). The results highlight the importance of providing nutritional knowledge and food literacy, developing nutritional skills and abilities to encourage consumers to make healthy food choices, and to strengthen other policies such as front-of-package labelling.