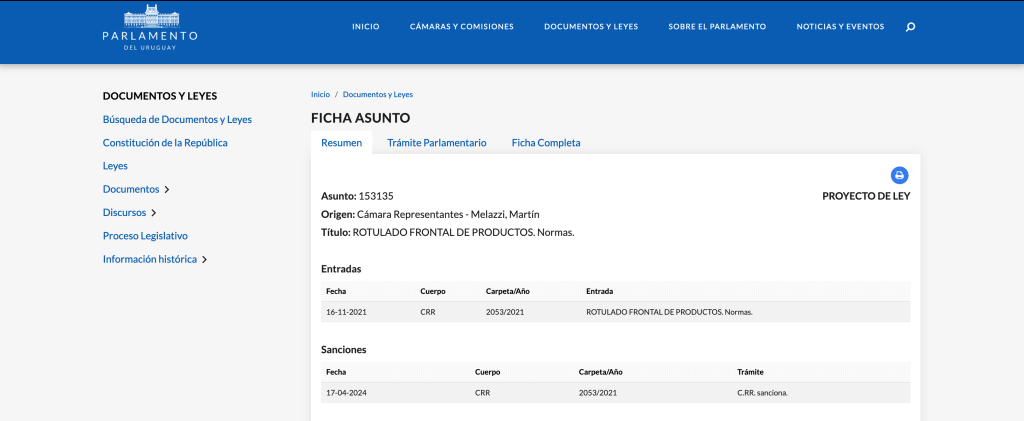
Marcela Reyes and María Luisa Garmendia, professors at INTA of the Universidad de Chile and CIAPEC researchers, were the researchers responsible for the FONIS “Consumption of non-caloric sweeteners in infants, preschoolers and adolescents of medium-low income after the implementation of Law 20.606”. In this article, they provide the available evidence to support the proposal of the Ministry of Health to implement a warning legend on packaged foods containing non-caloric sweeteners.